Hip Innovation Technology Product Testing
Reverse HRS Bench Level Tests
Extensive Bench Level testing has been completed on the Reverse HRS and additional research is ongoing. Bench Level research to date has included standard and uniquely designed tests. Many of these tests were designed to aggressively challenge a completely assembled Reverse HRS system. The testing categories included:
- Dynamic Fatigue
Durability testing conducted under active or simulated load
- Static
Stationary testing to verify structural design criteria, structural integrity and effects of limit loads
- Tribology
Wear testing of surfaces in relative motion
The Reverse HRS demonstrated excellent performance with all testing challenges including tribology, durability, and validation of structural design criteria, structural integrity and effects of limit loads. Some important testing results have been included for your review.
- Tribology: Polyethylene Wear Testing in Malposition
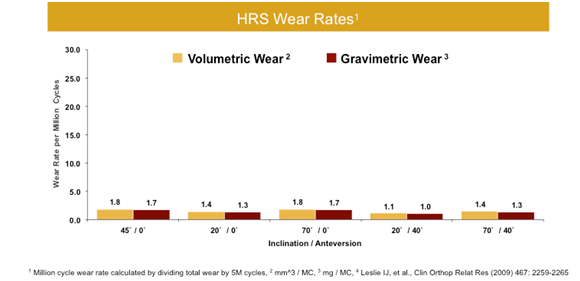
Standard wear testing was completed at "optimal" acetabular cup positioning of (45° inclination / 20° anteversion). In addition, modified testing occurred with a series of mal-positioned acetabular cups to simulate average annual polyethylene wear. The wear results were extremely positive and consistent regardless of acetabular cup placement, even in extreme malposition. These data further validate a primary product benefit that the Reverse HRS design has uncoupled the relationship between acetabular cup placement and component wear. Performance results in chart below:
Tribology: Polyethylene Wear Testing with Gait Separation
Gait Separation (evaluation of wear debris generation if there is loss of contact between the acetabular and femoral components of the system during the swing phase of the walking cycle) analysis was primarily performed for three important reasons:
- Ascertain polyethylene wear performance at various mal-positions
- Insure no occurrence of “runaway” polyethylene wear (runaway wear is a spike in wear rates that is not consistent with "steady state" wear rates)
- Determine if metal-on-metal (MoM) contact was possible between the acetabular cup and the femoral stem
It was once again determined that regardless of acetabular cup positioning, either in “optimal” or extreme malposition’s, the polyethylene cup wear was consistent and no “runaway” wear or edge loading occurred.
At inclination and anteversion positioning of 45°/0°, 20°/40° and 70°/40° the Reverse HRS was tested up to 10.5mm of separation during normal gait movement (walking) with no MoM contact. Extreme component positions of 20°/0° were terminated at 10.0mm and 70°/0° was stopped at 8.0mm with no MoM contact.
To summarize, at both normal and extreme mal-positioned component placement, the Reverse HRS tested well beyond the average separation of 1.3-1.5mm and the maximum separation reported in the literature of 5.2mm.

